Explain What the Different Colors in a Line Spectrum Represent
Light outside of this range may be visible to other organisms but cannot be perceived by the human eye. The electromagnetic spectrum can be expressed in terms of energy wavelength or frequency.
Continuous Spectrum Vs Line Spectrum Continuous Spectrum
The colours of the rainbow microwaves ultraviolet radiation and x-ray are some examples.

. What is the difference between a continuous spectrum and a bright-line emission spectrum. As plant vigor decreases the vegetation appears as lighter shades of red and pink various shades of greens and possibly tans. Bare soils appear as shades of white blue or green in most agricultural regions.
Their main difference is that continuous spectrum contains no gaps whereas line spectrum contains many gaps. What do the colors in a line spectrum represent. Red is also a color of Christmas bringing joy warmth and safety.
By this way the color produced is different from other elements. It can symbolize desire power speed and strength. If the gases used are not at low pressure there will be a continuous range of colours.
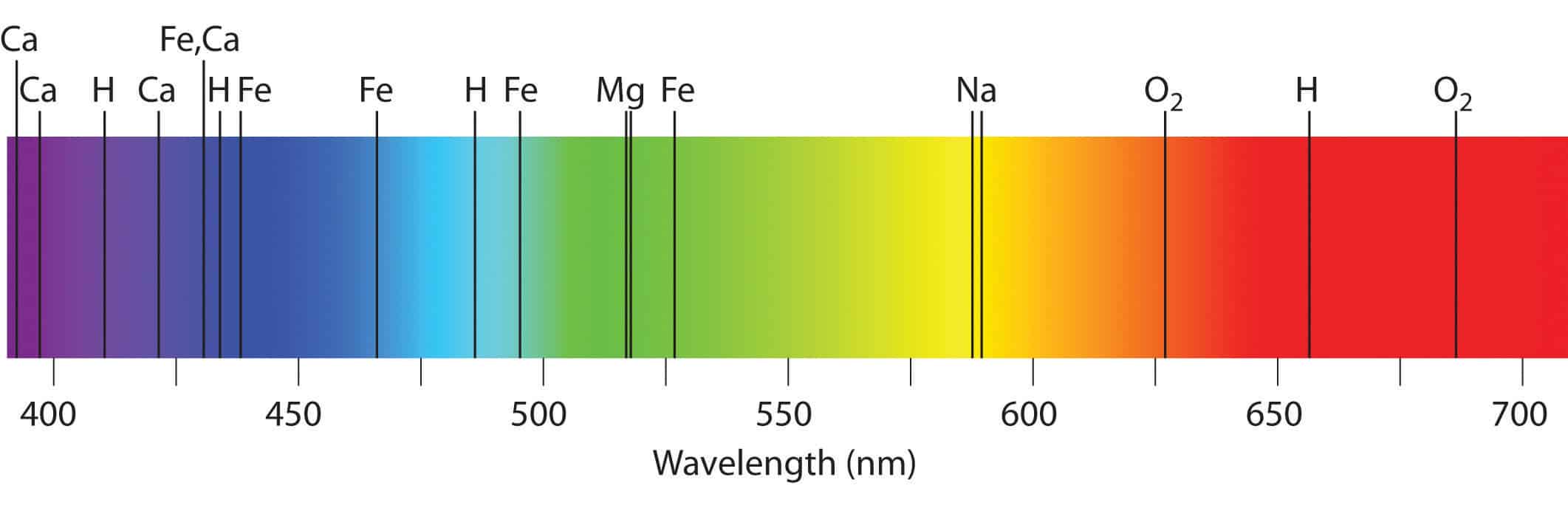
These observed spectral lines are due to the electron making transitions between the energy levels in an atom. The different colours in a line spectrum represent different frequencies of light. Bohr had an answer.
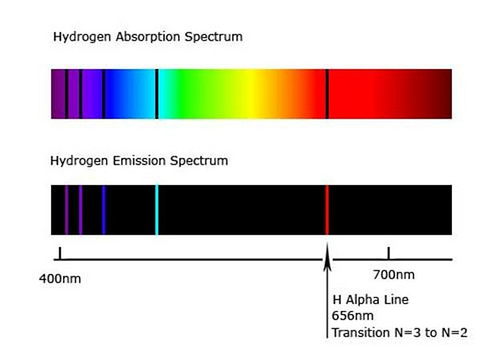
They represent the wavelengths of light that is absorbed by the spectrum They represent the energy that the atom gives off when their electrons are in place. When atoms are excited they emit light of certain wavelengths which correspond to different colors. A bright line emission spectrum shows only the colors that are associated with and emitted from a substance.
At high pressure tightly packed gas atoms or molecules will be vibrating rotating or colliding with each other such that many more energy levels will be created. Because each element has a different set of emission colors from the emission spectrum. On the other hand it stands for aggression danger violence and war.
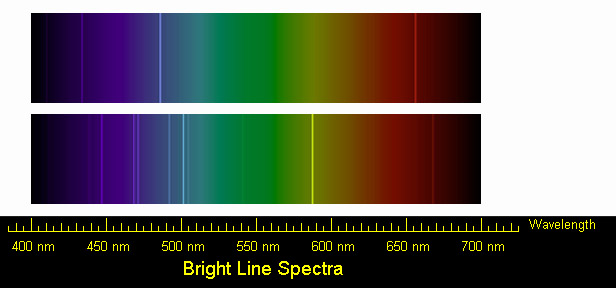
In general they convey optimism enthusiasm and passion. Transitions in each atom and the energy levels in it photons of different wavelengths and thus different colors are released from each gas. A continuous spectrum shows all the colors of the rainbow.
This spectrum of colors always appears in the same order. Each element produces its own unique line spectrum. The lighter the colour will represent light being BOUNCED off a.
Very intense reds indicate dense vigorously growing vegetation. The marks that you make on the three color lines will represent the different wavelengths of the different colors of light. Recorder divides the red line every 14 cm with dark marks in red pencil.
Red orange yellow green blue indigo and violet. When light passes through gas in the atmosphere some of the light at particular wavelengths is scattered resulting in. Each way of thinking about the EM spectrum is related to the others in a precise mathematical way.
Emission lines refer to the fact that glowing hot gas emits lines of light whereas absorption lines refer to the tendency of cool atmospheric gas to absorb the same lines of light. Because each element has a different set of emission colors from the emission spectrum. In general darker shades of each color indicate moister soil.
The different colors in a line spectrum represents when an electron falls from one energy level in an atom to a lower energy level it emits a photon of a particular wavelength and energy. These colors evoke warmth due to their brightness and link to the sun. Colors of light that correspond to narrow wavelength bands monochromatic light are the pure spectral colors learned using the ROYGBIV acronym.
These colors are considered cool as they are colors commonly found in nature and are known for their calming effect. You dont see all the wavelengths. A spectrum is characteristic of the elements present in the material considered.
The green line should be divided every 10 cm and the violet every 8 cm. All of the visible light wavelengths. Continuous spectrum and line spectrum are two types of spectra.
Because of this a line emission spectrum is like the fingerprint of the element which we can use to identify the element. The emission spectrum of hydrogen is divided into a number of spectral lines with wavelengths given by the Rydberg formula. Line emission spectrum.
Many of us consider red as a color of love but in fact it is more related to the excitement of falling in love and awaking passion for the other person. The color wheel is an illustrative diagram that shows 12 colors around a circle used to represent each colors relationship to one another. They represent the wavelengths of light that is absorbed by the spectrum They represent the energy that the atom gives off when their electrons are in place.
There are two color ranges that are not visible to our eyes in this spectrum. Colors arranged opposite each other are complementary to one another. Scientists represent wavelength and frequency by the Greek letters lambda λ and nu ν.
The true wavelengths are actually measured in terms of angstroms. What do the different colors in a line spectrum represent Why are the spectra for each element unique. Each element has its own unique line spectrum and is thus referred to as the fingerprint for a particular element.
White light is believed to contain all the seven rainbow colors which are absorbed at different wavelengths when refracted in a prism. Using those symbols the relationships between energy. No two gases give the same exact line spectrum.
Different Line of Spectrum As it says that the elements work by different energy level the energy level then produces different line of spectrum. Cool colors These include green blue purple and their variations like violet. If all the seven colors are present with no gaps between them it makes a continuous spectrum.
Below red is infra-red and above violet is ultra-violet. What do the individual lines in a bright line emission spectrum represent. You can see this color spectrum Red Orange Yellow Green Blue Indigo Violet and all the colors in between when you look through a diffraction grating.
So why do energized hydrogen gas molecules produce a line spectrum but not a continuous spectrum. Colors located near other colors share common characteristics and often pair well together. The lights travel on different line of.

Formation Of Spectral Lines Astronomy
Student Worksheet Graphing Spectra
Absorption Spectrum Emission Spectrum Lines Article Khan Academy
Comments
Post a Comment